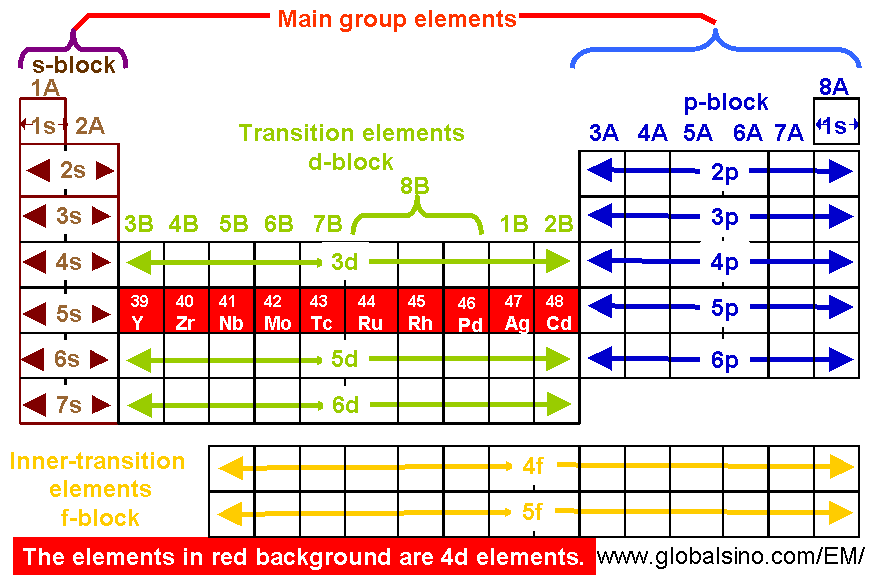
The eighth member of the period, neon, is almost totally inert. Proceeding through the period, the elements become less electropositive until the seventh member is the strongly electronegative fluorine. In the first period, the first member, lithium, loses an electron very easily – in other words, it is strongly electropositive. In each period there was a gradual change in properties from one element to its neighbor. When the elements were arranged in this way, they formed natural families. The next period of eight elements starts with sodium and ends with argon. These are followed by the period of eight elements starting with lithium and ending with neon. The two lightest elements, hydrogen and helium, form a short period of their own. He found that if the elements were arranged in order of increasing atomic weight, they fell into natural periods. This was eventually done, with outstanding success, by Dmitri Mendeleyev when he devised his periodic classification. Since the earliest days of chemical science, attempts had been made to put some sense and order into the classification of the elements – to explain the wide differences in the properties of some elements and the similarities of others. The properties of the elements differ greatly, ranging from highly reactive gases such as fluorine and chlorine and highly reactive metallic solids such as sodium and potassium to the inert gases and the heavy unreactive metals such as silver and gold. There are 92 naturally occurring elements and almost two dozen heavier, artificial elements. Than the first row, and stable in higher valence states. The second and third row transition elements are less reactive Many stable organometallic compounds and carbonyls (compounds in whichĬarbon monoxide, CO, acts as a ligand) with specially stable "push-pull"īonding. Properties: they exhibit many different valence states, form stable ligand complexes, are Their electronic structures, with many loosely-bound unpaired d electrons, account for their General the transition elements are dense, hard, and of high melting Short groups in the periodic table – i.e., groups IB through VIIB and group VIII – in which the d- orbitals are being filled. 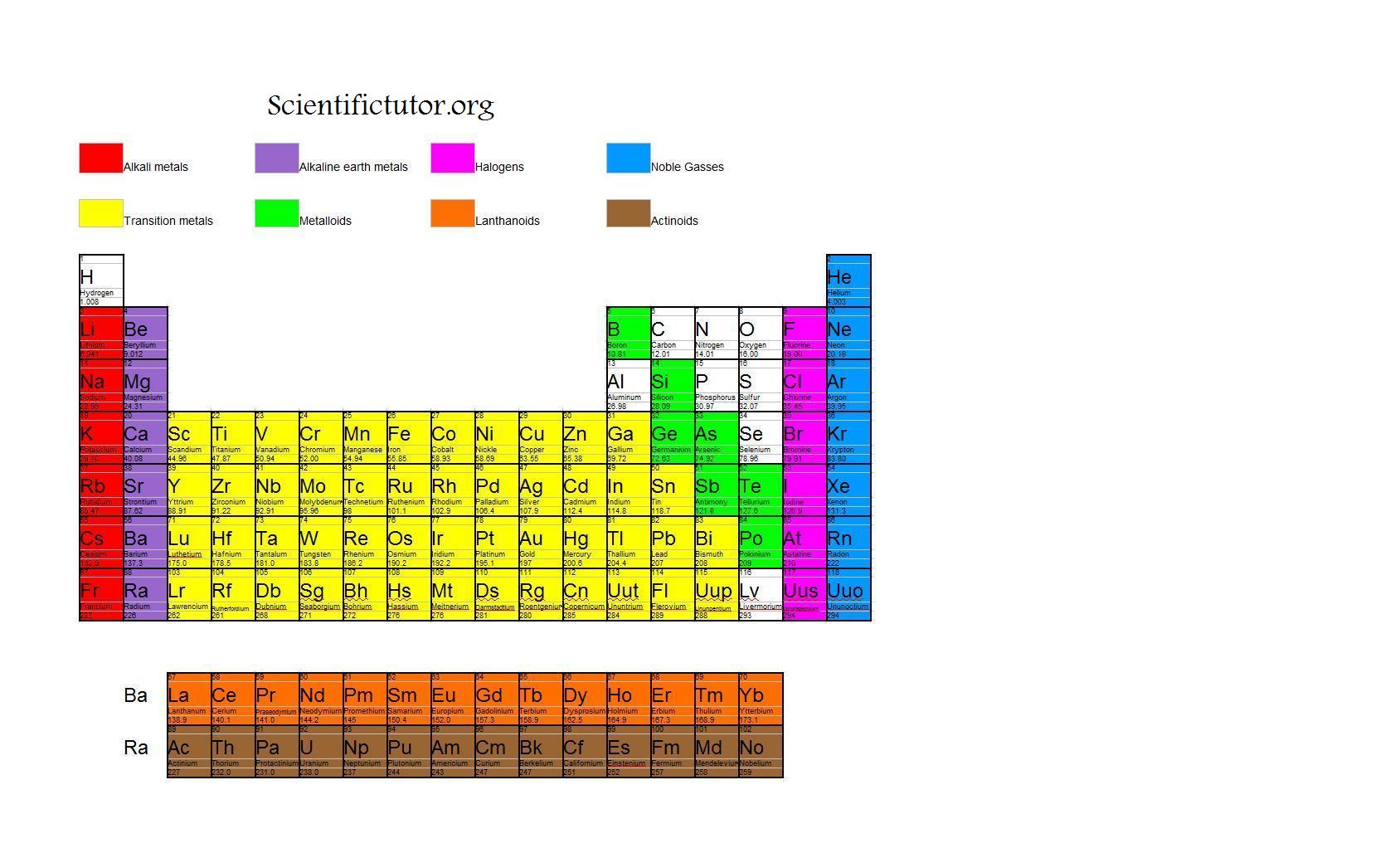
A transition element is a metallic element that has an incomplete
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
